 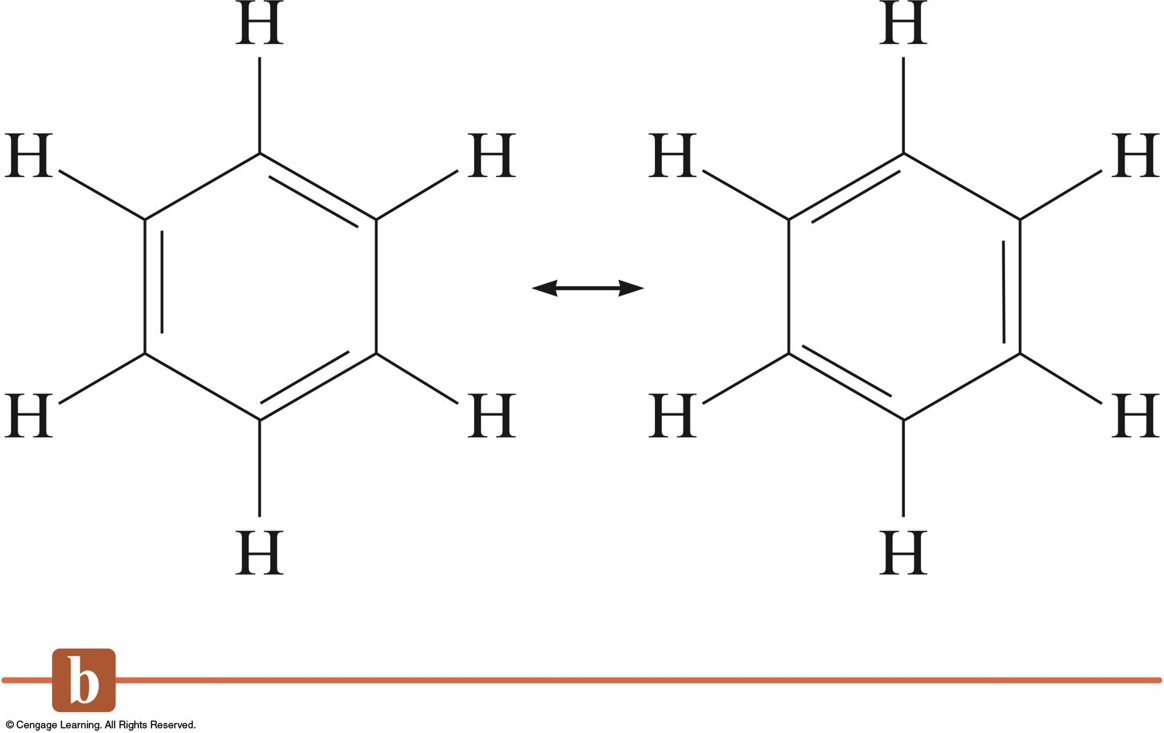
Hydrogenating something takes a lot of work. This reaction is a lot more sluggish than bromination, though. Hexene, CH 2=CHCH 2CH 2CH 2CH 3, could react with hydrogen to form hexane, CH 3CH 2CH 2CH 2CH 2CH 3. Hydrogen can add across double bonds to form "saturated hydrocarbons" (hydrocarbons that don't have any double bonds). As a result, the red-brown color of bromine disappears instantly if it is added to an alkene, because the bromine is rapidly consumed.Īlkenes also react with hydrogen, H 2. Ethene, or CH 2=CH 2, would add bromine to form dibromoethane, BrCH 2-CH 2Br. Bromine, Br 2 adds easily across alkene double bonds. For example, there are reactions that alkenes undergo pretty easily, but benzene does not. In some ways, aromatics behave differently than other compounds with C=C bonds. When we do that, we are saying that the real molecule behaves a little like one Lewis structure and a little like the other.ĭraw two Lewis structures that together illustrate the delocalization in benzene. We can try to show that benzene's bonds are all the same by using two resonance structures. Despite what the Lewis structure says, all of benzene's C-C bonds are the same length.They are not alternating, double-single-double as the Lewis structure suggests. The C-C bonds in benzene appear to be somewhere between a double and a single bond, and they are all the same. By comparison, a typical C-C bond is 1.47 Angstroms long, and a typical C=C bond is 1.35 Angstroms long. A C-C bond in benzene is 1.40 Angstroms long. This experiment allowed Lonsdale to measure the distances between the carbons in benzene. The first X-ray crystal structure of benzene was performed in 1929 by Kathleen Lonsdale. One of the pieces of experimental evidence that benzene has delocalized bonding comes from crystal structures. It is something that could be studied using a Huckel MO approach. These benzene units are sometimes called "phenyls" or, more generally, "arenes".īenzene is a good example of a delocalized structure. It is so common that it has gotten a great deal of attention over the years. 
Lots of compounds in nature contain "benzene units" within larger structures. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
